Why Are Host Cell Proteins Important in Biopharmaceutical Production?
Host cell proteins (HCPs) play a critical role in biopharmaceutical production. Their presence can impact product quality and safety. According to industry reports, HCP contamination can lead to adverse immunogenic reactions, compromising therapeutic efficacy. A study by the BioProcess International found that nearly 70% of biopharmaceutical developers reported significant challenges associated with HCP control.
The removal of HCPs during purification processes is essential. However, traditional methods often struggle to achieve the required purity levels. Recent innovations, such as affinity chromatography and advanced filtration techniques, have shown promise. Despite this, achieving optimal HCP clearance remains a complex and often costly endeavor.
Biomanufacturers continue to face the challenge of balancing yield and purity. Fluctuations in host cell metabolism can lead to variability in HCP levels. This variability necessitates rigorous testing and monitoring. As the industry evolves, increased understanding of HCPs and their influence on product performance is crucial. A focus on minimizing HCPs can significantly enhance the overall quality and safety profiles of biopharmaceuticals.

The Role of Host Cell Proteins in Biopharmaceutical Manufacturing
In biopharmaceutical manufacturing, host cell proteins (HCPs) play a critical role. These proteins result from the cells used to produce therapeutics. They can impact the efficacy and safety of the finished products. Understanding HCPs is essential for ensuring product quality.
The presence of HCPs can lead to unintended consequences. They may trigger immune responses in patients. This response can reduce the effectiveness of the treatment or cause adverse effects. Therefore, removing HCPs during purification processes is vital. Yet, this can be challenging, as not all purification methods can effectively eliminate them.
Developing reliable assays to measure HCP levels is also important. These assays must be sensitive enough to detect low levels of HCPs. However, current methods can lack precision. Scientists must continually refine these techniques to improve outcomes. The variability in HCP expression further complicates this issue. Continuous research is necessary to address these challenges in biopharmaceutical production.
Why Are Host Cell Proteins Important in Biopharmaceutical Production? - The Role of Host Cell Proteins in Biopharmaceutical Manufacturing
| Dimension | Importance | Impact on Biopharmaceuticals |
|---|---|---|
| Protein Purity | Ensures product safety and efficacy | Higher purity reduces risk of immune reactions |
| Yield | Influences overall production efficiency | Improved yield lowers production costs |
| Product Quality | Directly affects therapeutic performance | Quality variations can lead to therapeutic failures |
| Regulatory Compliance | Required for approval in pharmaceutical markets | Non-compliance can result in severe penalties |
| Stability | Influences shelf life and storage conditions | Enhanced stability improves patient access |
Understanding the Sources of Host Cell Proteins in Bioprocessing
Host cell proteins (HCPs) can be both a blessing and a curse in biopharmaceutical production. Understanding their sources is crucial for developing effective bioprocessing strategies. HCPs originate from the cells used to produce biopharmaceuticals. These cells include bacteria, yeast, and mammalian cells. When producing therapeutic proteins, HCPs can co-purify. This can create challenges during purification stages.
To mitigate HCP contamination, it’s essential to have a robust monitoring system in place. Regular assays can help detect HCP levels early on. This proactive approach can prevent costly downstream problems. A common tip is to utilize affinity chromatography during purification. It specializes in minimizing HCP presence, ensuring a cleaner product.
Additionally, be prepared for variability. HCP levels can change based on cultivation conditions. Factors like temperature, pH, and media composition can all play a role. Thus, consistent process controls are vital. Remember, even minor adjustments can lead to significant shifts in HCP contamination. Pay close attention to these details for successful biopharmaceutical development.
Sources of Host Cell Proteins in Biopharmaceutical Production
Impact of Host Cell Proteins on Product Quality and Safety
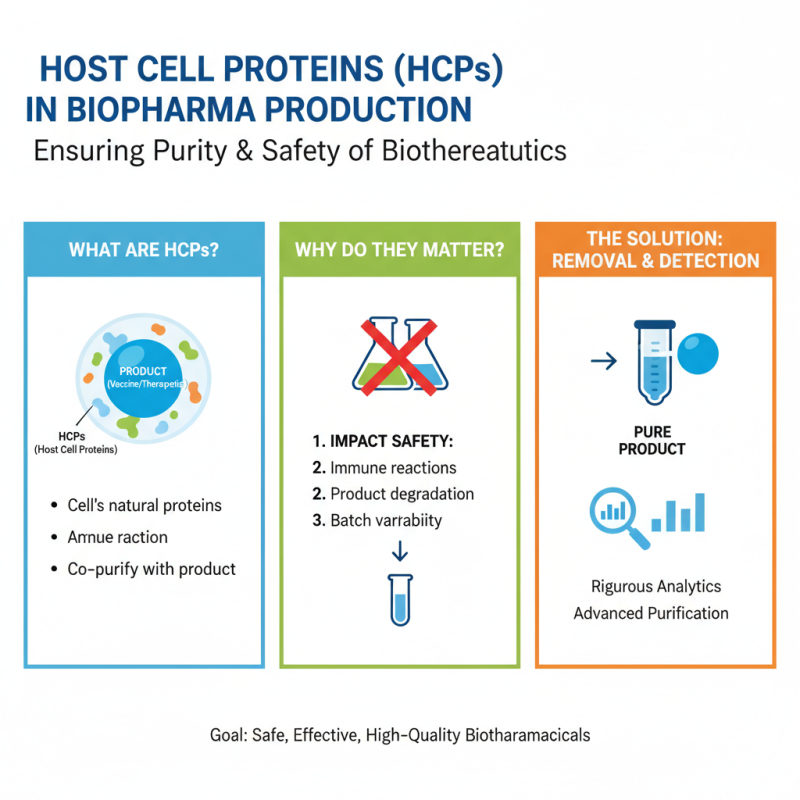
Host Cell Proteins (HCPs) play a critical role in biopharmaceutical production. These proteins are naturally present in the host cells used to produce vaccines and therapeutics. During production, HCPs can co-purify with the desired product. This co-purification can impact the safety and quality of the final biopharmaceutical.
The presence of HCPs might elicit an immune response in patients. Even trace amounts can lead to serious adverse effects. For instance, some patients may experience allergic reactions, which can complicate treatment regimens. Regulatory agencies stress the necessity of monitoring HCP levels throughout production. High levels of HCPs are not acceptable. Effective purification methods are essential but can be challenging. Each bioprocess varies in its ability to remove these impurities, which leads to inconsistencies.
Quality assessments must include thorough analysis of HCPs. These assessments often reveal gaps in the purification processes. Continuous improvements and new technologies are needed in this area. Inherent variability in host cells poses challenges to achieving uniformity. Small changes can lead to unforeseen consequences. Understanding these nuances is crucial for ensuring patient safety and product reliability.
Techniques for Analyzing and Characterizing Host Cell Proteins
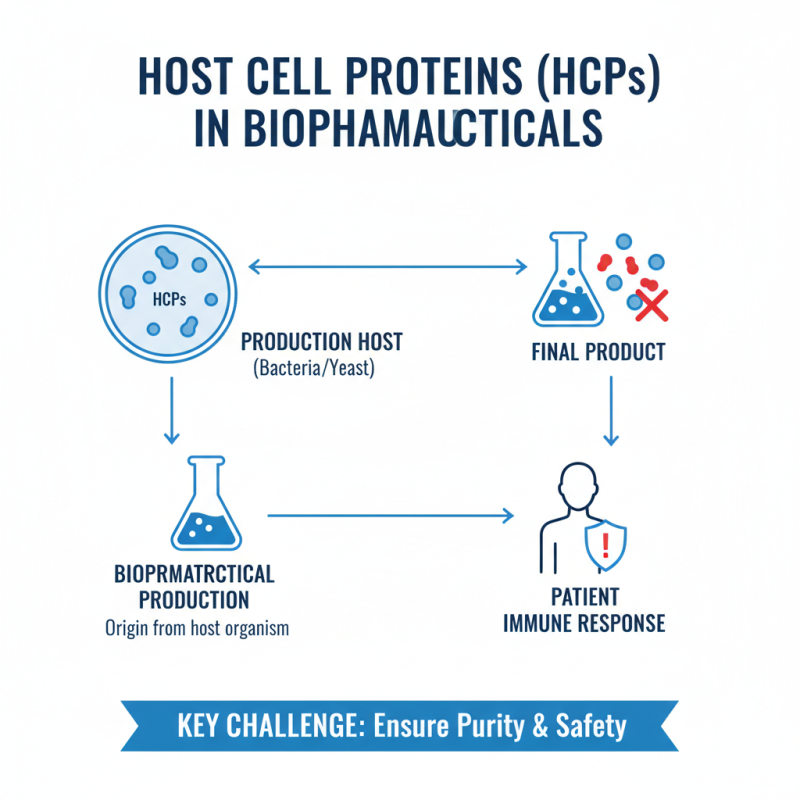
Understanding host cell proteins (HCPs) is vital in biopharmaceutical production. These proteins can originate from the production host, such as bacteria or yeast. If not properly controlled, HCPs may contaminate final products. They can trigger immune responses when administered to patients.
Techniques for analyzing HCPs have advanced significantly. One commonly used method is ELISA, which offers high specificity and sensitivity. This technique detects HCPs in trace amounts. Another valuable approach is mass spectrometry, enabling detailed characterization of protein profiles. Reports indicate that up to 50% of biopharmaceuticals may contain residual HCPs, making their analysis vital for patient safety.
Tip: Regularly validate your analytical methods. Use control samples to ensure accuracy. Employ multiple techniques for a comprehensive assessment of HCPs. This approach minimizes risk in drug development.
Additionally, human error in sample preparation can affect results. Proper training for lab personnel can reduce these mistakes. Invest in ongoing education to enhance skill sets related to HCP analysis. It can lead to more reliable data and foster better product safety.
Strategies for Reducing Host Cell Proteins in Biopharmaceuticals
Host cell proteins (HCPs) can significantly impact
biopharmaceutical production. These proteins may cause unwanted immune responses in patients.
Controlling HCP levels is crucial for ensuring product safety and efficacy.
High levels of HCPs can alter drug formulations and affect therapeutic outcomes.
To reduce HCPs, several strategies can be employed.
First, optimizing the host cell culture conditions can help. This includes adjusting nutrient levels and temperature.
By fine-tuning these parameters, the growth of unwanted proteins can be minimized.
Additionally, purification methods play a vital role. Techniques such as chromatography can selectively separate HCPs from the target protein.
However, these processes can be complex and costly.
Another approach involves using cell lines with lower HCP expression.
However, this requires thorough evaluation. Sometimes, these cell lines might not produce the desired yield.
In practice, balancing yield and HCP reduction is challenging.
Reflection on these strategies highlights that while we strive for purity, achieving it may come with trade-offs.
Each decision can impact the production process in unexpected ways.
Related Posts
-

Why Are Host Cell Proteins Important in Bioprocessing?
-

10 Essential Tips for Successful Protein Expression Techniques?
-
Top 10 Techniques for Protein Expression and Purification Explained
-

2026 How to Understand Gene Therapy and Its Potential Benefits?
-

Top 10 Benefits of Tissue Culture for Modern Agriculture and Horticulture?
-

How to Detect Mycoplasma in Cell Cultures Effectively?
