Why Are Host Cell Proteins Important in Bioprocessing?
In bioprocessing, host cell proteins (HCPs) play a crucial role. They are contaminants produced during the manufacturing of biopharmaceuticals. According to recent data from the BioPharmaceutical Consortium, 80% of manufacturing defects stem from HCP interference. This statistic highlights the need for robust control measures.
Dr. Sarah Jensen, a leading expert in protein purification, emphasizes, “Minimizing HCP levels is essential for ensuring product safety and efficacy.” Her insights reflect industry concerns. HCPs can trigger immune responses. They can impact the effectiveness of therapeutic proteins, such as monoclonal antibodies.
The challenge remains: completely eliminating HCPs is nearly impossible. Even with advanced purification techniques, residual HCPs can persist. This reality requires ongoing monitoring. It also demands innovative solutions in bioprocess development. As the demand for biologics rises, addressing HCPs becomes more critical than ever in ensuring patient safety.

Importance of Host Cell Proteins in Bioprocessing
Host cell proteins (HCPs) play a critical role in bioprocessing. These proteins can originate from the host organisms used for producing biopharmaceuticals. They may be found in recombinant proteins or therapeutic antibodies. Their presence can impact the quality of the final product. High levels of HCPs can lead to immune responses in patients. This poses a significant challenge for manufacturers.
Monitoring HCP levels is essential throughout the bioprocess. This process requires advanced analytical techniques. Detecting and quantifying HCPs early can minimize potential risks. Often, the removal of HCPs after product purification is incomplete. Striking the right balance between yield and purity is no easy task. Manufacturers must shift their focus to developing effective purification strategies.
Reduction of HCP content can drastically improve product safety. However, achieving this often complicates the overall process. Resource allocation becomes a challenge. More time and assets are required for rigorous testing. Companies may find themselves grappling with trade-offs between efficiency and safety. Potential contamination issues linger, making it crucial to reassess standard practices regularly.
Why Are Host Cell Proteins Important in Bioprocessing? - Importance of Host Cell Proteins in Bioprocessing
| Dimension | Description | Impact on Bioprocessing |
|---|---|---|
| Immunogenicity | Potential to trigger immune responses in patients | Can affect safety and efficacy of therapeutic proteins |
| Product Yield | Influence on the quantity of desired protein produced | High levels can dilute product yield or complicate purification |
| Purification Challenges | Presence of non-target proteins complicates downstream processing | Increased costs and time for purification processes |
| Regulatory Compliance | Requirement for characterization and control of impurities | Critical for meeting health authority guidelines |
| Process Optimization | Understanding host cell behavior can lead to improved processes | Enhances efficiency and productivity in biomanufacturing |
Role of Host Cell Proteins in Product Quality and Consistency
Host cell proteins (HCPs) play a critical role in bioprocessing. They can significantly impact product quality and consistency. HCPs are introduced during the production of biologics. Their presence can lead to unwanted immune responses. This is a concern for therapeutic protein development. According to industry studies, up to 70% of drug candidates fail due to quality issues related to HCPs. It highlights the importance of monitoring these proteins throughout production.
The removal of HCPs is essential for ensuring product safety. Advanced purification methods can reduce HCP levels considerably. This enhances the overall quality of the final product. However, complete removal is often not achievable. It’s a delicate balance between efficiency and effectiveness. HCPs can influence stability and bioactivity. Thus, understanding their impact is vital for maintaining consistency in bioprocessing.
Tips: Regular testing for HCPs is key. Implement robust analytical methods to monitor their levels. It can save time and resources. Staying updated on best practices in purification is equally important. Engage in continuous learning. The field evolves, and so should your processes. Never overlook the need for robust data to support decisions.
Impact of Host Cell Proteins on Biopharmaceutical Purification Processes
Host Cell Proteins (HCPs) play a critical role in the purification of biopharmaceuticals. During the production of these drugs, cells used in fermentation or cell cultures release various proteins. These proteins can interfere with the desired product. Higher levels of HCPs can complicate the purification process.
Managing HCPs is challenging. Traditional purification methods may not fully remove these contaminants. This may impact product yield and purity. In some cases, high HCP levels can lead to adverse immune responses in patients. This highlights the need for thorough HCP removal and monitoring during purification.
Effective strategies are essential for dealing with HCPs. Modern techniques, such as affinity chromatography, are commonly employed. Yet, such methods may not capture all unwanted proteins. Researchers must continuously refine their approaches. Regular assessments and validations are necessary for optimal results. Each bioprocessing situation may require unique solutions.
Impact of Host Cell Proteins on Biopharmaceutical Purification Processes
This chart illustrates the impact of Host Cell Proteins on four key metrics in biopharmaceutical purification processes: Process Yield, Cost, Time, and Purity. Understanding these impacts is crucial for optimizing bioprocessing workflows.
Strategies for Managing Host Cell Proteins during Biomanufacturing
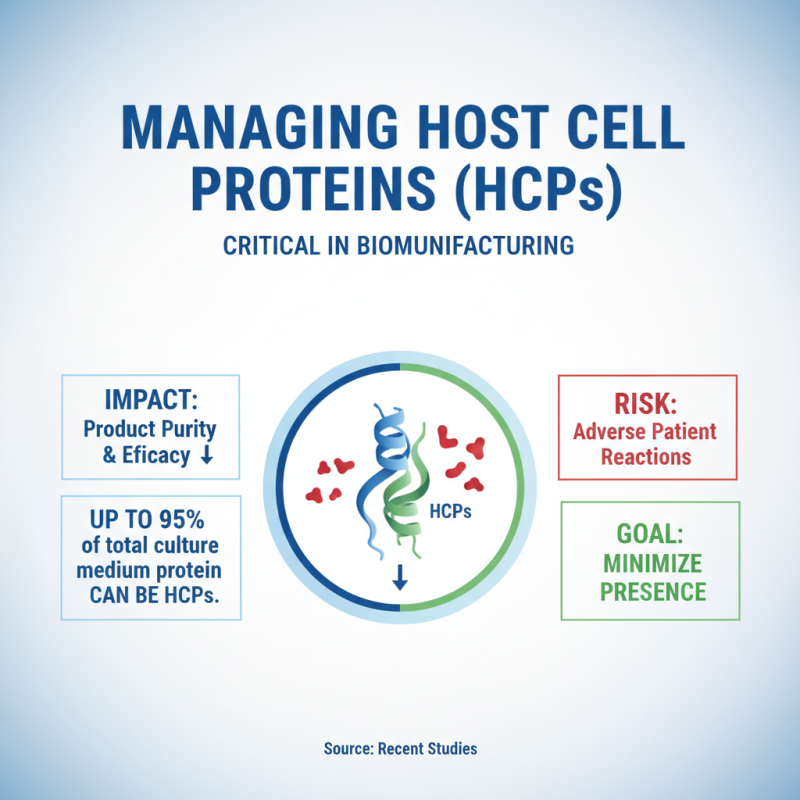
Managing host cell proteins (HCPs) is critical in biomanufacturing. HCPs can impact product purity and efficacy. According to recent studies, up to 95% of the total protein in the culture medium can be HCPs. These proteins might cause adverse reactions in patients. Therefore, their presence must be minimized.
One strategy involves using affinity chromatography techniques. This method selectively removes HCPs while preserving target proteins. In a recent report, this approach reduced HCP levels by more than 90%. Another tactic is optimizing culture conditions. Adjusting parameters like pH and temperature may minimize HCP production, but it can also impact protein yield. Thus, careful monitoring is essential.
Despite advances, challenges remain. HCP detection methods vary, leading to inconsistent data. Some proteins are also similar in structure to therapeutic proteins, complicating purification. The industry must focus on refining protocols. Continuous research on HCP characterization and removal is needed to enhance product safety and efficacy. The journey toward optimal bioprocessing involves ongoing reflection and improvement.
Future Perspectives on Host Cell Proteins in Bioprocess Optimization
Host cell proteins (HCPs) play a critical role in bioprocessing. They can affect the quality of biopharmaceuticals. High levels of HCPs can lead to immunogenicity issues. Therefore, understanding HCPs is vital for future drug development. As we refine our processes, optimizing the removal of HCPs becomes paramount.
Looking ahead, bioprocess optimization will need innovative strategies. New purification techniques are emerging, yet they are not flawless. Some methods may still leave behind residual HCPs. We must evaluate the trade-offs between yield and purity. This balance can be tricky. The industry is still learning how to achieve this ideal. Collaboration between researchers and manufacturers could help in finding new solutions.
Investing in advanced analytics will also be essential. It allows for better detection and characterization of HCPs. However, the technology is not yet fully developed. Continued efforts are needed to make these tools accessible. Understanding HCPs will remain a significant challenge, but addressing this issue is critical. We must keep pushing the boundaries for safer and more effective biopharmaceuticals.
Related Posts
-

10 Essential Tips for Successful Protein Expression Techniques?
-
Top 10 Techniques for Protein Expression and Purification Explained
-

2026 How to Understand Gene Therapy and Its Potential Benefits?
-

Why Are Recombinant Proteins Important for Biotechnology and Medicine?
-

How to Understand Gene Therapy and Its Impact on Modern Medicine?
-

2026 Top Trends in Life Sciences Innovations and Discoveries?
