10 Essential Tips for Understanding Host Cell Proteins in Biotechnology
Understanding host cell proteins (HCPs) is crucial in biotechnology. These proteins are unintended by-products of the production process. They can impact the quality and safety of biopharmaceuticals.
HCPs are derived from the cells used to produce therapeutic proteins. Their presence can lead to immunogenic responses in patients. This makes it essential to monitor and control HCP levels. Various techniques are employed to detect and quantify these proteins. Yet, challenges remain in fully eliminating them from final products.
Researchers often face difficulties in characterizing HCPs. The diversity and complexity of these proteins add to the challenge. Not all HCPs have known functions, making them unpredictable. Addressing these issues is vital to enhance product development and patient safety. Understanding HCPs requires both careful study and continuous improvement.

Overview of Host Cell Proteins in Biotechnology
Host cell proteins (HCPs) play a crucial role in biotechnology, particularly in the production of biopharmaceuticals. These proteins, derived from the host organisms used for expression systems, can contaminate the final products. Understanding HCPs is essential for ensuring product safety and efficacy. According to a report by BioPharma, over 80% of biopharmaceutical products experience issues related to HCP contamination. This makes it vital for biotechnologists to grasp the significance of HCPs.
One key tip for managing HCPs is to implement robust purification processes. Employ various techniques such as affinity chromatography and ion exchange. These methods can help reduce HCP levels significantly, often by more than 95%. This reduction is essential for meeting regulatory standards. Another important aspect is to conduct thorough analytical testing. Identifying HCPs early in the development can mitigate risks later in the production.
Training staff on the implications of HCPs is often overlooked. Providing continuous education can strengthen awareness and instill a culture of quality. According to industry surveys, only 30% of staff are well-informed about HCP-related issues. Awareness can lead to improved practices and better outcomes in biomanufacturing. Fostering an environment that encourages open discussions about HCP challenges is vital for progress. Understanding and tackling these complexities is crucial for successful bioprocessing.
Importance of Monitoring HCP Levels in Biopharmaceutical Production

Monitoring host cell proteins (HCP) is crucial in biopharmaceutical production. HCPs can impact product quality, safety, and efficacy. According to industry reports, approximately 70% of biopharmaceuticals contain residual HCPs from production host cells. This presence can trigger immune responses in patients, making it imperative to monitor HCP levels closely.
Effective HCP monitoring can prevent costly recalls and ensure compliance with regulatory standards. A study by the Biotechnology Industry Organization noted that 30% of product failures in clinical stages were linked to unexpected HCP levels. These failures indicate a pressing need for continuous evaluation throughout the manufacturing process, not just at the end. The industry has yet to standardize methods, causing variability in results. Such inconsistency raises questions about the reliability of our data.
Moreover, the tools for HCP detection and quantification are evolving. Current techniques, including ELISA and mass spectrometry, can provide insights, yet they often don't perform well across different platforms. This reflects the challenge in ensuring robust detection methods that can adapt to varied bioprocessing environments. While we have advanced, the path forward demands reflection and improvement in HCP monitoring practices.
Techniques for Identifying and Characterizing Host Cell Proteins
In biotechnology, understanding host cell proteins (HCPs) is crucial. These proteins can interfere with the efficacy and safety of biopharmaceuticals. Identifying and characterizing HCPs require robust techniques. For instance, mass spectrometry is widely used. It provides comprehensive analyses of complex protein mixtures. According to a report by BioPharma Research, over 70% of companies rely on mass spectrometry for HCP identification.
Another effective technique is enzyme-linked immunosorbent assay (ELISA). This method allows for the specificity of HCP detection. While it is efficient, ELISA can miss low-abundance proteins. In fact, a recent industry study revealed that 35% of HCPs could be overlooked when using ELISA alone. This highlights a significant gap in detection strategies.
The challenge lies in the diversity of HCPs. Variability depends on the host cell line and production conditions. Often, proprietary processes complicate the identification efforts. Moreover, the need for stringent regulations adds pressure. Companies must find a balance between thorough testing and resource management. The stakes are high, as undetected HCPs can lead to severe consequences, including product recalls. Thus, developing complementary approaches is vital for effective HCP characterization.
Regulatory Guidelines for HCP Assessment in Drug Development
Understanding Host Cell Proteins (HCP) is crucial in biotechnology, especially in drug development. Regulatory guidelines dictate strict assessment of HCP levels. The presence of HCP can affect drug safety and efficacy. The FDA outlines requirements for evaluating HCP in biologics. A recent report indicates that 50% of drug recalls are linked to HCP issues. It highlights the need for robust testing.
Each batch of biologics must undergo HCP analysis. The acceptable limits vary by product. Often, limits are set to less than 100 ppm for therapeutic proteins. However, achieving this can be challenging. Not all HCPs are equal. Some may elicit immune responses, complicating matters further. Developers face a constant balancing act. The need for speed in development clashes with thorough assessments.
Common techniques for HCP detection include ELISA and mass spectrometry. However, variability in results can exist. Different methods may yield different quantifications. This inconsistency raises questions about reliability. As industry standards evolve, ongoing education and adaptation are essential. Stakeholders must confront these issues head-on to ensure patient safety and product integrity.
10 Essential Tips for Understanding Host Cell Proteins in Biotechnology - Regulatory Guidelines for HCP Assessment in Drug Development
| Tip No. | Tip Description | Importance | Regulatory Reference |
|---|---|---|---|
| 1 | Understand HCP origins in recombinant protein production. | High | FDA Guidance |
| 2 | Implement robust analytical methods for HCP detection. | Critical | ICH Q6B |
| 3 | Conduct risk assessments of HCP based on product type. | Medium | EMA Guidelines |
| 4 | Regularly review manufacturing processes to minimize HCP contamination. | High | FDA Q7A |
| 5 | Document all changes in HCP monitoring protocols. | High | ICH Q10 |
| 6 | Establish high-level testing thresholds for HCPs. | Critical | FDA Guidelines |
| 7 | Engage in thorough training for personnel on HCP issues. | Medium | Guideline Reference |
| 8 | Continuously monitor product development for HCP trends. | High | EMA Q8 |
| 9 | Utilize in-silico methods to evaluate HCP risk. | Medium | FDA Innovations |
| 10 | Keep abreast of evolving regulations on HCP assessment. | Critical | Ongoing Regulatory Updates |
Strategies for Reducing HCP Contamination in Bioprocesses
Host Cell Proteins (HCPs) are a significant concern in biotechnology. They can contaminate biological products during production. Effective strategies to minimize HCP contamination are crucial for ensuring the safety and efficacy of biopharmaceuticals. Recent industry reports indicate that over 80% of biologics are affected by HCP presence, impacting their regulatory compliance.
One effective approach is optimizing purification processes. Techniques such as affinity chromatography and ion exchange can reduce HCP levels significantly. For instance, utilizing a multi-step purification workflow can decrease HCP contamination rates to as low as 1%. However, the challenge lies in the potential loss of product yield during these processes. Balancing purity and yield remains a complex task for biomanufacturers.
Implementing analytical technologies is also vital. Advanced techniques like mass spectrometry can enhance HCP detection and quantification. Recent studies suggest that they can identify over 90% of HCPs. However, these methods require sophisticated equipment and trained personnel, which can be costly. The challenge often lies in the scalability of these technologies for large-scale production. Ultimately, continuous innovation is needed to navigate these complexities in bioprocessing.
Related Posts
-

Why Are Host Cell Proteins Important in Bioprocessing?
-

Why Are Host Cell Proteins Important in Biopharmaceutical Production?
-
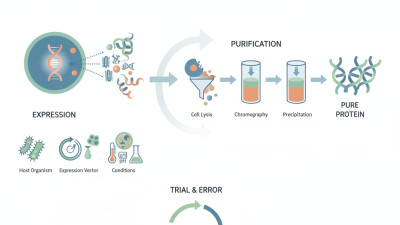
What is Protein Expression and Purification in Biotechnology?
-

10 Essential Tips for Successful Protein Expression Techniques?
-
Top 10 Techniques for Protein Expression and Purification Explained
-

2026 How to Understand Gene Therapy and Its Potential Benefits?
